The decision to legalize cannabis for recreational use has been a boon to the industry, enabling states across America to open up new markets. This newfound freedom of access has pushed businesses in this sector into an innovation arms race as they strive for consumer attention and loyalty. Delta 8 products like gummies, vape cartridges, cookies, capsules, oils and tinctures have cropped up all over the place- there’s no shortage of ways people can get their hands on some THC!
The main takeaway here is that while we’re lucky enough now to enjoy leisurely marijuana consumption legally in many US States – it pays off hugely when consumers prioritize quality control before any purchase; you really don’t want subpar cannabinoids running through your veins!.
Top 5 Delta 8 Carts In The Market 2023
-
Exhale Wellness – Overall Best Delta 8 Carts; Editor’s Pick
-
BudPop – Potent Delta 8 THC Vape Cartridges
-
Hollyweed CBD – Premium Quality Delta 8 Carts
-
Diamond CBD – Trusted Delta 8 Cartridges; Non-GMO Ingredients
-
3Chi – Best Delta 8 Carts With 100% Hemp-derived THC Oil
#1. Exhale Wellness – Overall Best Delta 8 Carts; Editor’s Pick

This is one of the top cannabis-producing brands in the US. They have their headquarters and facilities in Los Angeles. The team behind the effective running of the company is made up of farmers, researchers, and cannabis enthusiasts who feel the need to educate people on the benefits of cannabis and create products that are safe to use.
Exhale Wellness has built a reliable reputation of being one of the consistently manufactured top-quality delta-8 products. The brand has committed to creating its products with natural and organic ingredients that do not contain GMO or artificial ingredients. The flavors being offered for their delta-8 cartridges are all-natural, and they include Mango, blackberry, pineapple, and others.
The amount of delta-8 found in a hemp plant is minimal, so it is imperative that the extraction process employed is of top quality; the brand employs the CO2 extraction method – it is regarded as one of the best. In addition, they use isomerization to concentrate the delta 8. Exhale Wellness offers users great cartridges, and they also run battery replacement services.
Highlights
-
The source of the hemp is essential is how the final product will turn out. One of the best places to get quality hemp is in Colorado, where exhale source theirs from. They make their cartridges from natural and organic ingredients that promote usage by vegans. Their delta-8 does not contain dairy, artificial flavors, or gluten.
-
There is a limit of dosage set by the farm bill regarding cartridges. Exhale’s cartridges have a concentration of 900mg delta-8 in every milliliter. In addition, the brand ensures its products are tested by third-party labs to ensure their claims are valid and to put users at rest.
-
The brand has a reputation for putting the quality of its product and customers’ safety first; this reputation has held the brand down all these years. However, the brand has been covered by magazines such as Forbes, LA Weekly, the Observer, Los Angeles Times, and the ministry of hemp. All of this coverage has propelled the brand to new heights and projected the brand to users for the quality they offer.
-
Customers are catered for through the brand’s free shipping policy, and they offer users a Cashback policy on some of their purchases. Every first-time customer gets to enjoy a 20% off the total cost of their first purchase. Considering how many people love discounts, this has been an attraction tool for new users. The brand also has a policy of returning 100% of unsatisfied customers’ money when they return the product within thirty (30) days.
Pros
-
They offer users the chance to save money through the discounts on their platform, their money-back policy offers a great timeframe for return, and they ship orders to users for free.
-
Their products are made from all-natural and organic ingredients of the best quality to ensure user safety and comfort.
-
All their products are suitable for vegans.
-
The brand’s cartridges are compatible with the standard510 pens.
-
Their delta-8 cartridges are created without including harmful substances like PEG oil, VG, MCT, and PG.
-
The customer reviews about this brand are almost 100% positive.
Cons
=> Click here to visit the official website “Exhale Wellness”
#2. Budpop – Potent Delta 8 THC Vape Cartridges

For every milliliter of budpop’s D8 cartridges, there is a delta-8 concentration of 800mg; this is effective, especially for first-time users. Budpop has its offices and facilities in the United States, and they source their hemp from farms In Nevada.
Their cartridges come in two flavors: Grape Runts and strawberry gelato. The flavors are of two different strains – the strawberry gelato is of an indica strain, while the grape runtz is of a hybrid strain. Both flavors have a strong taste.
Budpop ensure that their product is created from ingredients that do not contain GMOs and are vegan friendly. These ingredients are checked for pesticides and other harmful chemicals.
The brand ensures that the hemp they use is cultivated through an ethical cultivation process, refined, and used to create delta-8 products in their GMP-certified facilities. The Budpop avoids the use of harmful substances such as Vitamin E, MCT, VG, PG, or PEG.
Highlights
-
The brand has a money-back guarantee timeframe of thirty (30) days. Unsatisfied customers have to return the product within this time frame to receive 100% of their money back. However, quick notification is encouraged by the brand.
-
The brand ensures that a certified third-party lab checks its products. These testing results (COAs) are posted on their website for customers to see. Customer satisfaction is guaranteed, and they try to make them feel comfortable through their several money-saving offers, such as a 20% discount for new customers. There are also discounts for ex and present military personnel and those with disabilities.
-
The brand’s insistence on quality products is essential to its growth. They obtain their hemp from farms in Nevada that follow an ethical cultivation process. They make their cartridges using quality materials such as stainless steel and food-grade silicone. This is safe for users to consume.
-
The brand’s delivery service is one of the fastest on the market; they process orders within one (1) or two (2) days and get them to the customer quickly. Unfortunately, the brand does not deliver products to certain states due to legalization restrictions.
Pros
-
They show their concerns for veterans and present military personnel by offering discounts.
-
Facilities certified by GMP create their products.
-
Their products are tested and verified by third-party labs.
-
The materials used in making their cartridges are durable and of top quality.
-
New customers are liable to enjoy a discount on their first-time purchases.
-
They ship orders for Free to the customer’s doorstep.
-
They offer a thirty (30) day money-back guarantee for unsatisfied customers.
Cons
=> Click here to visit the official website “BudPop”
#3. Hollyweed CBD– Premium Quality Delta 8 Carts

Hollyweed CBD has all its facilities and operations in California; they were established in 2017. The brand has enough experience to know that top-quality products are needed, and they have delivered.
The brand has a six-pillar motto that guides its operations; the spiritual, physical, intellectual, emotional, and social well-being of customers and the environmental impact of their delta-8 products. Hollyweed CBD has a delta-8 concentration of 900mg per cartridge.
They make their cartridge using natural ingredients/materials that do not include PG, MCT, PEG oil, and VG. The brand also avoids including delta-9 THC in its cartridges. Their products are great at offering users’ relaxation and a euphoric feeling. The brand’s cartridges are created in two forms; the push-to-activate and inhale-to-activate cartridges.
Highlights
-
The brand has been in the industry for a long time; this is an advantage over the new brands. Their experience in the market is valuable to creating quality products as they have had their trial-and-error phase. The brand had a head start on researching Ingredients and flavors that add more quality to a product. The team behind the brand’s running has a combined valuable experience in growing and producing quality delta-8 products that are safe to use and offer great health benefits.
-
The brand maintains its flavor varieties of blackberry, sour diesel, and Jack Herer flavors. The brand has also maintained its no cruelty and vegan-friendly products. In addition, they ensure their delta-8 cartridges go through third-party lab testing.
-
There are varieties to everything about the brand’s products. These delta-8 cartridges have a variety of flavors, and they come in different strains with separate Concentration levels. To enjoy the product as effectively as hoped, users are advised to follow the brand’s dosage inscription on the product’s container. In addition, Hollyweed CBD helps its customers earn money on its platform through its affiliate program. There is a wholesale program for those interested in joining the business to help them save money.
Pros
-
Their products are tested by a third-party lab and certified to be the highest quality.
-
The brand avoids using chemicals or artificial ingredients at any point in the manufacturing process. Instead, they ensure the ingredients/materials are 100% natural.
-
The brand offers users their cartridges in no less than ten (10) flavors.
-
They ensure their product is free of cruelty and also vegan-friendly.
-
They paste instructions on using the product for effective results on the container.
-
The brand’s cart effectively creates a soothing feeling that can help reduce the level of stress.
Cons
=> Click here to visit the official website “Hollyweed CBD”
#4. Diamond CBD – Trusted Delta 8 Cartridges; Non-GMO Ingredients

This company has its headquarters and facilities in Florida; it was established in 2015. The team behind the establishment and running of the brand is made up of experts from several professions, such as farmers, doctors, and scientists.
The driving force behind the brand’s success is its mission to develop new means to develop more ways for users to use delta-8 and the benefits they can enjoy. In addition, the brand is open to global reach.
Diamond CBD sources its hemp from several locations such as Scandinavia, Colorado, and Kentucky. The brand uses full-spectrum delta-8 strains, which means the hemp has to undergo an extraction process to extract the cannabinoids, terpenes, and other nutrients.
The band mixes their delta-8 extract with CBD, and they do this to enhance the euphoric feeling users experience and guarantee more membrane relaxation. In addition, Diamond CBD offers users an array of flavors such as banana, Mango, sour diesel, blueberry, strawberry, and others.
Highlights
-
The brand loves to put the concerns of their customers first, essentially why they have plenty of discount offers. At the first instance of creating an account and subscribing via the brand’s website, you will enjoy 70% off the total cost of your purchase. When you refer your friend or family to buy the brand’s products, you will get a $20 coupon. The company prides itself in creating products that leave an amazing aftertaste while tasting smooth while consuming. For all orders from $100 and above, the customers will enjoy free delivery with a delivery time estimate of two (2) days.
-
Customers that wish to ask for their money back have to return the product within thirty (30) days after purchase, and the product must be returned unopened. The brand ensures that all return orders are treated fairly and promptly. Alternatively, you will get an email response from their customer service team concerning your refund request within three (3) to seven (7) days of initiating the request. And within the time of the processing of your request, Diamond CBD will keep you updated.
-
The brand ensures its production facilities are of the top quality and that its products are all vegan friendly. All of the hemp extracts they use are free of GMOs, so they try to preserve the organic nature by not adding extra animal byproducts. The customers who have used their cartridges have earmarked their effectiveness towards improving their sleep, helping them eat better, and improving how they feel at the moment.
Pros
-
The brand’s combination of delta-8 and CBD makes their products more effective in giving that euphoric and relaxing feeling.
-
They ship all orders costing from $100 and above for free.
-
They offer customers packages that encourage them to buy and refer their friends.
-
They ensure their products get tested by a third-party lab that verifies all of the company’s claims.
-
The flavor on offer is a lot.
-
The customer review section on their website had over ten thousand (10,000) positive reviews.
-
The brand has a great social media presence with over fourteen thousand (14,000) followers on Instagram.
-
The brand obtains its hemp from farms that use the organic cultivation process.
-
The brand has been covered by some of the biggest magazines such as Yahoo Finance, The Courier, observer, LA weekly, and economies.
Cons
#5. 3chi – Best Delta 8 Carts With 100% Hemp-derived THC Oil

3Chi is one of the brands that focused on rightly blending in flavors with quality; most users enjoy this. As a result, their cartridges come in different flavors, offering different amazing tastes and cannabinoids. The team behind the brand’s running understands that one product can not be used to solve all problems. Hence, they segregated their delta-8 cartridges to cater to different times of the day, different moods, and different intentions.
This is one of the first brands to enter the delta-8 market when the use was legalized; this has contributed to the popularity amassed by the brand. Their products have been in line with the regulation limit of no more than 0.3% delta-9 THC concentration per product. They have successfully created quality products that have earned them a great brand reputation.
3chi has been consistent in using natural ingredients/materials to create its cartridges; their fifteen (15) years of research experience have helped maintain and create quality. The brand uses natural Terpenes as an agent to flavor their cartridges which leaves an amazing after-use taste in the mouth. Additionally, the brand encourages brand transparency by posting its certificate of analysis on its website for customers to see.
Highlights
-
The brand’s segregation technique has helped them. They have a cartridge meant for certain times of the day and specific feelings. Their cartridge varieties are wide, such as delta-8 cartridges mixed with HHC, or delta-8 cartridges mixed with THCP and THC-O. That is not all, and there are still more on offer; you can find them on the brand’s website. There is a cartridge for any mood you are feeling; either you want to feel calm and focused, or you want to feel happy or relaxed and at ease – you can get a cartridge for any of these feelings. The brand ensures that its vape liquid holds a high potency level and has a high concentration of delta-8.
-
The brand is focused on serving its customers and ensuring they are comfortable using its products and services. 3Chi offers customers who subscribe to their website various discounts and coupon offers with an amazing shipping fee. The brand’s team believes there are still a lot of benefits to be tapped from cannabis, and they are committed to discovering those benefits. The brand manufactures its cartridges using natural materials and avoids using thinning oils.
Pros
-
Their delta-8 cartridges are compatible with standard510 batteries.
-
They do not charge an excessive amount for delivery.
-
They have different types of cartridges that are meant for different moods.
-
They offer users different favoring types.
-
The brand focuses more on creating a safe to consume and quality product.
-
Users can enjoy discounts when they buy the products in bulk.
-
The ingredients used by the brand are of the best quality.
-
Their cartridges are durable as they are made from glass tanks and ceramic coils.
-
In each cartridge, there is a 1000mg concentration of delta 8.
Cons
-
The website does not fully talk about the brand’s history.
-
They do not offer quick shipping times; it could take up to five (5) days before users get their product.
-
Their vapes are meant for single use.
How We Made This List Of The Most Potent Delta 8 Cartridges
As a well-invested user of cannabis products, it could be a difficult adventure when you set out to try something new. It is more difficult for those starting their cannabis adventure.
The aim of all these is to ensure you can select a brand that will satisfy your needs without adding to your problem.
After researching the number of manufacturing brands for delta 8 carts on the market, we outlined some criteria that helped us throughout our selection process. This way, we can post an unbiased review of the best brands.
In addition, we ensured the brands we selected had top-notch quality, and customers enjoyed using them.
These are the criteria(s) we used:
-
Compliance With Federal Law:
In 2018, a farm bill was passed to guide the production of cannabis byproducts; any brand that did not align their products to this regulation was thrashed.
In addition, the bill stated that all cannabis-producing brands must ensure that their products do not contain more than 0.3% THC concentration.
The FDA served companies that did not comply with warning letters; we checked online for warnings received by these brands. We also cross-checked the third-party lab results on their websites.
The only brands of delta 8 carts that we were able to see results of their third-party lab testing are those that were transparent enough to post it on their websites. These results will show if the claims made by the company on ingredients, safety, potency, and effectiveness were true.
The FDA has several labs that have been approved to carry out third-party lab tests; however, some brands sideline those labs and do theirs with an inside lab – we ignored these brands. And also, it is possible to manipulate these results; these can be done through the third-party lab, so we called each lab to be sure of the results.
The source of the hemp used is also a piece of important information, and the results stated that. In addition, the type of product it is and if it contains harmful substances would be stated. We checked for these to ensure the product is of top quality.
A brand’s reputation is essential when buying new delta-8 carts. What other people are saying about the brand and what they are known for are what make up their reputation.
We checked this to ensure the brand was legit, and there were no complaints of severe side effects after use. Customer reviews helped us, and we also checked if any brand was covered in a magazine or a reputable blog.
It is essential that a brand is willing to sell to many users – this means they are willing to deliver to an array of locations. Understandably, some states are beyond reach due to their laws against cannabis products; however, how well can a brand get to the customers in the states that have legalized cannabis.
Some of these brands of delta 8 carts try to cater to customers’ needs as comfortably as possible; some provide free shipping of orders to the customer’s doorstep. And for those that did not offer that, we checked if the shipping fee was affordable, and the estimated delivery time is important.
The value you are getting for the price you are being asked to pay is essential; you have to remain within budget, which we recognize and approve of. Some brands charge exorbitant prices for their product, which end up being below par, so it is essential to ensure you are getting great value for money.
The size of the cartridge, the flavors being offered, and the level of the product’s potency is essential to creating value. These three (3) factors helped us slim down the list. We then used the ingredients to judge the brands left in making the product. The cartridge must be made for durable use because that represents the best value for money.
The effectiveness of the cartridge is essential in ensuring customer satisfaction. Customer reviews are the best place to learn about this, so we read many of them online and through review sites to know the brand that most customers complained about their ineffective products.
After that, we checked the brands left on our list and tried their products one after the other; this way; we had a first-hand experience of their effectiveness.
This is one of the factors that can build a brand from scratch to the top at a fast rate. When a brand is transparent about its affairs with its customers, it helps forge a loyal relationship between both parties; the customers will feel part of the manufacturing process, so they will be less skeptical about using the brand’s products.
The first step to being transparent is by posting your third-party lab results on your website so customers can check if the product is good for them. Also, brands can be transparent about manufacturing their delta-8 cartridges; the customers will understand the process and know it is safe for them to use.
We can not overstate how research can be helpful to brands who want to create a new product. Knowledge drives success: this is the only way you can create an efficient and safe product for consumption.
When brands research the ingredients required to make a top-quality product, they can channel that knowledge into creating one.
The top brands are not only concerned with creating a top-quality product, but they are also after improving the ones they have out in the market. The aim is to help delta-8 products get to new heights in the industry.
What makes up a product is the ingredients used in making it; it is of great value for a brand to use active, natural, and organic ingredients to produce their delta-8 cartridges.
All of the brands we recommended above were scrutinized to ensure they served users a product made from the best quality and safest ingredients.
Some products contain chemicals, so it is imperative to avoid such brands. Additionally, on its Ingredients list, users should avoid any brand with acidic precursors, vernolic acid, or p-cymene terpene. These ingredients can get into the cart’s content, thereby resulting in severe health issues for the user.
Buying Guide: Factors To Consider Before Buying Top-quality Delta 8 Carts
Several businesses have an opportunity to make money from the delta-8 industry; this has caused the birth of several brands.
However, not all these brands offer the quality products needed for effective use. We have listed some factors below to help you decide on the product you would love to use.
Price:
There is a façade that is passed around that the expensive products are of the best quality; however, this is false. The brand’s reputation determines the trueness of that. A brand that is well known for its quality can quote its product at a high price and still get users to buy it. However, you should first have a budget before searching for a product to buy; this will help you check on products you might not enjoy as much as you think. In addition, the product you choose must offer great value for your money.
Brand Name:
The brand’s name means you are buying your product from Matt a lot. Some brands are known for the quality product they produce consistently. If you want to patronize a new brand, you should scrutinize their website for valuable information about the quality of their products. The brand you patronize must-have details of their product on their website so you can use this information to decide if the product is quality enough for you.
The company owners matter a lot, the objective that guides their business operations, and their history in the same industry or one similar. Unfortunately, some brands are just out to make money without caring about the customer’s safety and satisfaction. Please select a brand that has a capable team running its affairs.
Source Of Their Delta-8:
The final product’s quality is almost determined by the quality of the hemp used in creating it. You should check where the brand sourced its hemp from as you want to purchase the product. Some brands are open about information of this kind; they state the source of their hemp on their website or on the body of the packaging.
Some locations are synonymous with cultivating quality hemp; ensure the brand gets their hemp from one of these locations. Ensure they do not use chemicals such as pesticides or GMOs during cultivation. If you are a vegan, check for their Ingredients to be sure it is vegan-friendly.
Manufacturing Methods:
You need to know how the product was manufactured. There is no stand-alone delta-8 THC vape cartridge; it has to be extracted from the hemp plant, so it is imperative to know the extraction method employed in getting the delta-8 out.
Certain extraction methods will get the good nutrients out of the hemp plants, thereby resulting in a quality product beneficial to you. Knowing how distilled the product is also important; the ingredients mixed with the delta-8 must be natural and organic.
The Types of Ingredients:
This is the aspect of your check that is most important and requires maximum attention; this is necessary, especially for a product that mixes with your blood. You can check for the ingredients list regardless of if you are buying the product online or offline.
When purchasing a product online, ensure the brand has the product’s Ingredients list posted along with it. And if you are buying offline at a store, you should ask the salesperson about the ingredients used in making the product.
This is essential, especially if you are buying offline at the store; they can have a scam or harmful product on the shelf, so it is essential to know the ingredients. Any cartridge brand Ingredient list you are checking must contain cannabinoids, cannabis-derived terpenes, and vegetable glycerine and glycerol—the terpenes in the ingredient list act as a natural flavoring agent.
Product’s Potency:
The potency of the product you want to buy should match what you need; this is essential for your maximum satisfaction. As a beginner, you should start with a low potency level before walking up the ladder. As for those doing delta-8 cartridges for a while, you can choose a potency level that works for your system.
Product’s Availability:
Product availability is essential to check. Some products are of top quality, but they are mostly sold out due to the high demand for such products. And some products look impressive, but the brand that creates that product is no more in business.
So, do not get excited when you find a product you think will suit your needs; try to find out if it is available before getting your hopes high. The location also matters; some brands do not ship to countries outside of the United States or some states in the US. You should check all this to ensure the product is available to you.
Customer Friendliness:
Selecting a brand focused on satisfying its customer will help you get a product of the highest quality. Also, check for a brand with responsive customer service and how well they serve their customers.
You should make inquiries to gauge if the customer service agents are good enough for you. Finally, check for discounts and coupon offers, free shipping policy, and return policy to know how well they look to make their customers comfortable.
FAQs About Delta 8 Carts
Q1: How Do Delta-8 Cartridges Work?
A cartridge contains a liquid concentrated with delta 8; a battery is charged and used to power the cartridge. Some have a button you push to initiate the healing of the liquid inside the cart, which will result in vapors. This vapor is what you will inhale.
Q2: Is Using Delta-8 Cartridges Legal in the US?
According to federal law, it is legal to use delta-8 products with a concentration of no more than 0.3% delta-9 THC. However, some states have rejected that legalization and maintained that using delta-8 products is illegal.
Q3: Is It Possible to Get High from Delta-8 Cartridges?
This is possible, considering users tend to feel something euphoric after using it. CBD products are the ones that do not get a user high. However, the high feeling will not be as intense as when consuming delta 9.
Q4: What Is the Correct Dosage Level for Delta-8 Cartridges?
There is a prescription for how you should consume delta-8 carts. The dosage you apply to consume delta-8 depends on your body’s metabolism. Your level of tolerance will guide your dosage level. However, for beginners, you should start your dosage level low, then you can increase it if you feel your tolerance level can handle it.
Q5: What Is the Best Way to Store Delta-8 Cartridges?
You have to keep your cartridge in a cool place away from sunshine. The heat of the sun can burn out the potency of the product. We advise you to ensure your cartridge is properly sealed after use and ensure your cartridge is kept away from air and moisture.
Q6: Is It Possible to Pass a Drug Test After Consistently Consuming Delta-8 Cartridges?
This is quite unlikely. Whether you are consuming delta-8 or delta-9, the testing machine can not differentiate if one is legal or illegal because they both contain THC. However, you can test negative for a THC test if your body processes the nutrients faster.
Conclusion: Recommended Quality Brands For Delta 8 Cartridges
The essence of this article was to recommend quality brands that manufacture delta-8 cartridges. We have recommended five (5) brands so you can have options to choose from as their products do not offer the same benefits.
If you are after the potency, flavor, or any other feature, all the brands we have listed above can provide those. They are mostly affordable, with amazing shipping and discount offers. As a Beginner, it might be better for you to go with our recommendations to ensure you are enjoying maximum quality.
 The index fell 23.7% in Q3 and 13.8% in Q4, ending 2022 down 70.4%:
The index fell 23.7% in Q3 and 13.8% in Q4, ending 2022 down 70.4%: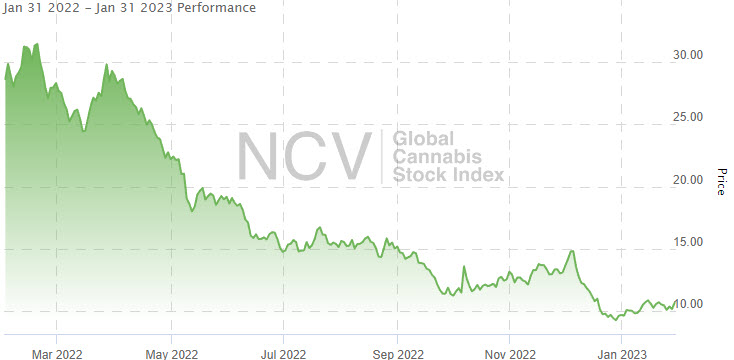 Since the peak in February 2021, the Global Cannabis Stock Index has dropped 88.4% from the 92.48 closing high:
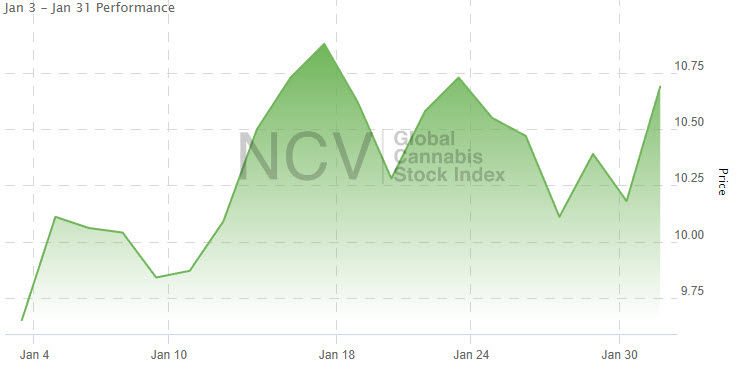
Since the peak in February 2021, the Global Cannabis Stock Index has dropped 88.4% from the 92.48 closing high: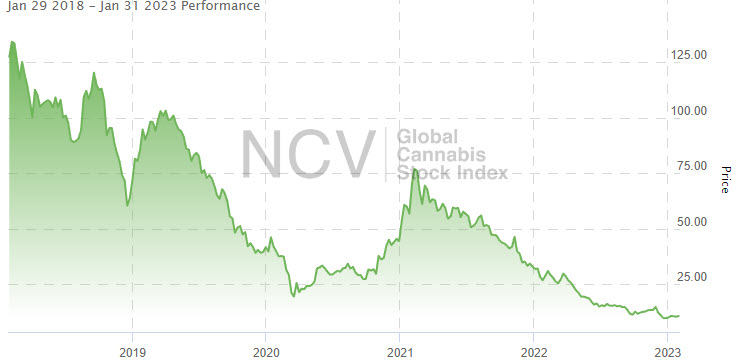 The strongest 4 names in January all rose by more than 31%:
The strongest 4 names in January all rose by more than 31%: Get ahead of the crowd by signing up for 420 Investor when it becomes available again. It’s the largest & most comprehensive premium service for cannabis investors since 2013.
Get ahead of the crowd by signing up for 420 Investor when it becomes available again. It’s the largest & most comprehensive premium service for cannabis investors since 2013.

